Abstract
Introduction: Ciltacabtagene autoleucel (cilta-cel), a chimeric antigen receptor T (CAR-T) cell therapy with 2 B-cell maturation antigen (BCMA) targeting single-domain antibodies, demonstrated early, deep, and durable responses in the phase 1b/2 CARTITUDE-1 study in patients (pts) with relapsed/refractory multiple myeloma (RRMM) who had been heavily pretreated (Berdeja, Lancet, 2021). After a median follow-up of 12.4 months, the overall response rate (ORR; as assessed by independent review committee) was 97%, with 67% of pts achieving stringent complete response (sCR). The 12-month progression-free survival (PFS) and overall survival (OS) rates were 77% and 89%, respectively. Here, we report updated results from CARTITUDE-1 with longer duration of follow-up (median 18 months).
Methods: Eligible pts had MM and received ≥3 prior therapies or were refractory to a proteasome inhibitor (PI) and an immunomodulatory drug (IMiD), and had received a PI, IMiD, and an anti-CD38 antibody. After apheresis, bridging therapy was permitted. Pts received a single cilta-cel infusion (target dose 0.75×10 6 CAR+ viable T cells/kg; range 0.5-1.0×10 6) 5-7 days (d) after lymphodepletion (300 mg/m 2 cyclophosphamide, 30 mg/m 2 fludarabine daily for 3 d). Primary objectives were to characterize cilta-cel safety, confirm the recommended phase 2 dose (phase 1b), and evaluate cilta-cel efficacy (phase 2). Response was assessed per IMWG criteria by an independent review committee and minimal residual disease (MRD) negativity at 10 -5 by next-generation sequencing. In this combined phase 1b and phase 2 analysis, cytokine release syndrome (CRS; per Lee et al Blood 2014 grading criteria) and neurotoxicity (by CTCAE v5.0) were mapped to the ASTCT criteria for CRS and immune effector cell-associated neurotoxicity (ICANS), respectively.
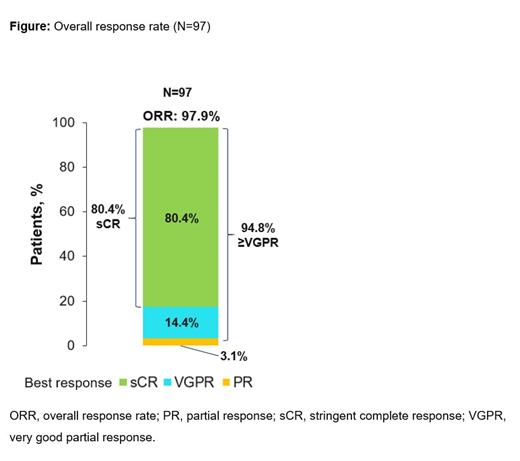
Results: As of Feb 11, 2021, 97 pts (58.8% male; median age 61.0 years [range 43-78]) received cilta-cel. Pts had received a median of 6 (range 3-18) prior lines of therapy; 83.5% were penta-drug exposed, 87.6% were triple-class refractory, 42.3% were penta-drug refractory, and 99.0% were refractory to last line of therapy. ORR was 97.9% (95% CI: 92.7-99.7); 80.4% of pts achieved sCR, and 94.8% achieved very good partial response or better (Figure). The median time to first response was 1 month (range 0.9-10.7), median time to best response was 2.6 months (range 0.9-15.2), and median time to complete response or better was 2.6 months (range 0.9-15.2). The median duration of response was 21.8 months (95% CI: 21.8-not estimable). Of 61 pts evaluable for MRD, 91.8% were MRD negative at the 10 -5 threshold; MRD 10 -5 negativity was sustained for ≥6 months in 44.3% (27/61) of pts and ≥12 months in 18% (11/61) of pts. The 18-month PFS and OS rates were 66.0% (95% CI: 54.9-75.0) and 80.9% (95% CI: 71.4-87.6), respectively. 18-month PFS rates in pts who achieved sustained MRD for ≥6 months and ≥12 months were 96.3% (95% CI: 76.5-99.5) and 100%, respectively. The most common grade 3/4 hematologic AEs in ≥25% of pts were neutropenia (94.8%), anemia (68.0%), leukopenia (60.8%), thrombocytopenia (59.8%), and lymphopenia (49.5%). There were no fatalities related to cytopenias, and no new safety signals with longer follow-up. CRS occurred in 94.8% of pts (mostly grade 1/2); median time to onset was 7 d (range 1-12), and CRS resolved within 14 d in 98.9% of pts. There was no new CAR T-cell neurotoxicity since the previous report. Post cilta-cel infusion, 21 deaths occurred during the study: 0 within first 30 d, 2 within 100 d, and 19 more than 100 d post infusion. Ten deaths were due to disease progression, 6 were treatment-related (as assessed by the investigator), and 5 were due to AEs unrelated to treatment. One patient was retreated with cilta-cel (for progressive disease) and had stable disease (per computerized algorithm) post-retreatment with no incidence of CAR T-cell neurotoxicity.
Conclusions: At a longer median follow-up of 18 months, a single cilta-cel infusion led to early, deep, and durable responses in heavily pre-treated pts with MM. Follow-up is ongoing and updated data will be presented. Cilta-cel demonstrated a manageable safety profile with no new safety signals observed with longer follow-up. Further investigations of cilta-cel are ongoing in earlier lines of therapy (CARTITUDE-2 [NCT04133636], CARTITUDE-4 [NCT04181827], and CARTITUDE-5 [NCT04923893]) and in outpatient settings.
Martin: GlaxoSmithKline: Consultancy; Janssen: Research Funding; Sanofi: Research Funding; Amgen: Research Funding. Usmani: Takeda: Consultancy, Research Funding, Speakers Bureau; SkylineDX: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; Merck: Consultancy, Research Funding; Janssen Oncology: Consultancy, Research Funding; Bristol-Myers Squibb: Research Funding; EdoPharma: Consultancy; Array BioPharma: Consultancy, Research Funding; Celgene/BMS: Consultancy, Research Funding, Speakers Bureau; GSK: Consultancy, Research Funding; Sanofi: Consultancy, Research Funding, Speakers Bureau; Janssen: Consultancy, Research Funding, Speakers Bureau; Amgen: Consultancy, Research Funding, Speakers Bureau; Abbvie: Consultancy. Berdeja: Lilly, Novartis: Research Funding; Poseida, Sanofi, Teva: Research Funding; GSK, Ichnos Sciences, Incyte: Research Funding; Bluebird bio, BMS, Celgene, CRISPR Therapeutics, Janssen, Kite Pharma, Legend Biotech, SecuraBio, Takeda: Consultancy; Celularity, CRISPR Therapeutics: Research Funding; Abbvie, Acetylon, Amgen: Research Funding; EMD Sorono, Genentech: Research Funding. Jakubowiak: Amgen: Membership on an entity's Board of Directors or advisory committees; GSK: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Gracell: Membership on an entity's Board of Directors or advisory committees; Sanofi: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees. Cohen: Novartis: Research Funding; BMS/Celgene: Consultancy; Takeda: Consultancy; Genentech/Roche: Consultancy; Oncopeptides: Consultancy; AstraZeneca: Consultancy; Janssen: Consultancy; GlaxoSmithKline: Consultancy, Research Funding. Hari: Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Karyopharm: Consultancy; Sanofi: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Oncopeptides: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Millenium: Membership on an entity's Board of Directors or advisory committees, Research Funding; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Adaptive Biotech: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau; Celgene-BMS: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding, Speakers Bureau. Avigan: Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Pharmacyclics: Research Funding; Kite Pharma: Consultancy, Research Funding; Juno: Membership on an entity's Board of Directors or advisory committees; Partner Tx: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Aviv MedTech Ltd: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Legend Biotech: Membership on an entity's Board of Directors or advisory committees; Chugai: Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy; Parexcel: Consultancy; Takeda: Consultancy; Sanofi: Consultancy. Deol: Kite, a Gilead Company: Consultancy. Lesokhin: bristol myers squibb: Research Funding; Iteos: Consultancy; pfizer: Consultancy, Research Funding; Behringer Ingelheim: Honoraria; Trillium Therapeutics: Consultancy; Serametrix, Inc: Patents & Royalties; Genetech: Research Funding; Janssen: Honoraria, Research Funding. Munshi: Adaptive Biotechnology: Consultancy; Karyopharm: Consultancy; Oncopep: Consultancy, Current equity holder in publicly-traded company, Other: scientific founder, Patents & Royalties; Legend: Consultancy; Novartis: Consultancy; Abbvie: Consultancy; Celgene: Consultancy; Janssen: Consultancy; Takeda: Consultancy; Amgen: Consultancy; Pfizer: Consultancy; Bristol-Myers Squibb: Consultancy. O'Donnell: Onocopeptide: Consultancy; Karyopharm: Consultancy; Janssen: Consultancy; Bristol Myer Squibb: Consultancy; Adaptive: Consultancy; Takeda: Consultancy. Stewart: Oncopeptides: Honoraria; Janssen: Honoraria; GSK: Honoraria; BMS: Honoraria; Amgen: Honoraria; Skyline diagnostics: Consultancy; Genomcs England: Membership on an entity's Board of Directors or advisory committees; Tempus Inc.: Current holder of individual stocks in a privately-held company, Membership on an entity's Board of Directors or advisory committees; PikSci Inc.: Current holder of individual stocks in a privately-held company, Patents & Royalties; Sanofi Aventis: Honoraria. Schecter: Janssen: Current Employment, Current holder of stock options in a privately-held company. Goldberg: Janssen: Current Employment, Current holder of individual stocks in a privately-held company. Jackson: Janssen: Current Employment; Memorial Sloan Kettering Cancer Center: Consultancy. Yeh: Janssen: Current Employment. Banerjee: Janssen: Current Employment, Current holder of individual stocks in a privately-held company. Allred: Janssen: Current Employment, Current holder of individual stocks in a privately-held company, Current holder of stock options in a privately-held company. Zudaire: Janssen: Current Employment. Deraedt: Janssen: Current Employment. Olyslager: Janssen: Current Employment. Pacaud: Legend Biotech: Current Employment. Lin: Juno: Consultancy; Bluebird Bio: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; Novartis: Consultancy; Takeda: Research Funding; Kite, a Gilead Company: Consultancy, Research Funding; Vineti: Consultancy; Gamida Cell: Consultancy; Sorrento: Consultancy; Legend: Consultancy; Merck: Research Funding; Janssen: Consultancy, Research Funding. Jagannath: Legend Biotech: Consultancy; Karyopharm Therapeutics: Consultancy; Janssen Pharmaceuticals: Consultancy; Bristol Myers Squibb: Consultancy; Sanofi: Consultancy; Takeda: Consultancy.
At the time of abstract submission, cilta-cel is being investigated for the treatment of multiple myeloma but is not yet approved.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal